
Research
Highlighted results
The development of Adaptos® technology is based on high-quality research. Discover the science behind Biomendex innovations through these highlighted results.
Excellent bone-material contact
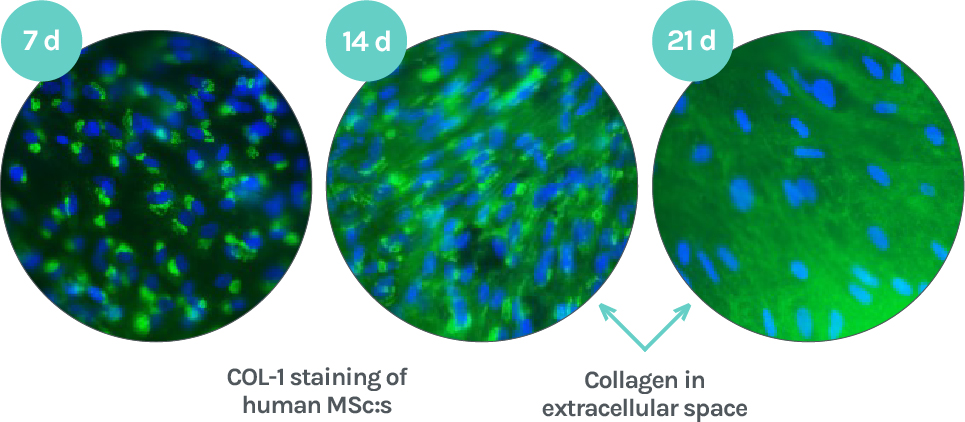
Adaptos® composite has been shown to support the viability, proliferation, and osteogenesis of human adipose tissue derived stem cells in vitro. Especially the formation of a collagenous mineralized extracellular matrix is a strong indication of osteogenesis in a cell culture environment. (See publication 2)
The safety of Adaptos® composite has also been shown in a rabbit femur and cranial defect models. Moreover, these studies demonstrated that bone tissue attached directly to bone and bone was seen to grow on Adaptos® surface and to infiltrate into the porous structure already after four weeks of implantation. (See publications 1, 2)
Optimal pore structure
A high interconnected porosity (native bone 60–90 %) with a pore size of 150–400 µm are crucial for bone tissue and vascularisation regeneration and ingrowth. Microcomputed tomography-based methods have been evaluated in the analysis of Adaptos® composites (see publication 3) and applied to deeper understanding and validation of Adaptos® structure.

Resorption rate
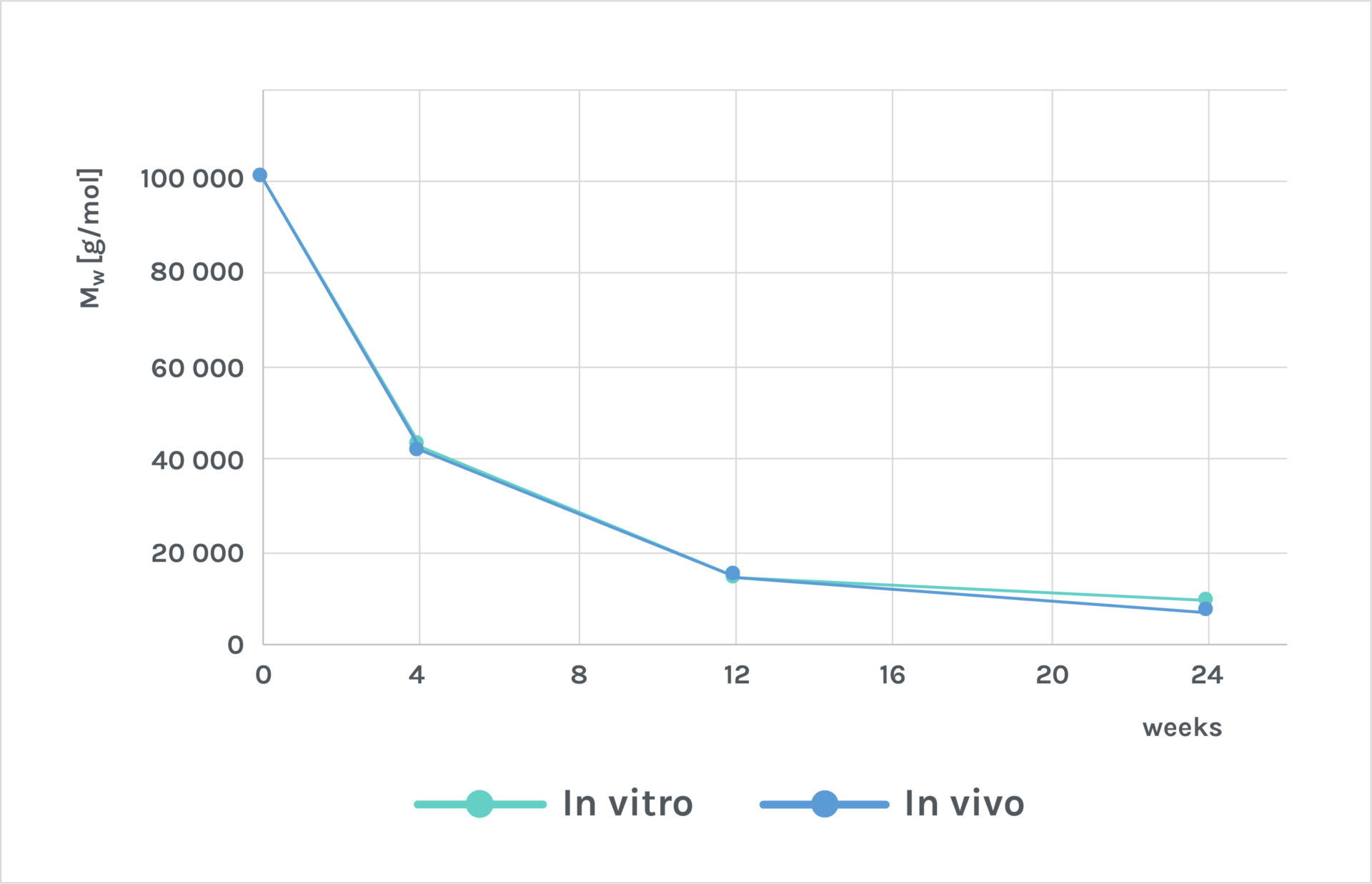
Adaptos® bone grafts are designed to fill a bone defect, and support and facilitate natural bone regeneration while resorbing away as the native tissue heals. Adaptos® bone substitutes are composed of fully absorbable components that degrade into molecules and ions, which also normally appear in the body and are secreted via natural metabolic routes. The degradation of Adaptos® composite has been shown to degrade in a controlled manner both in vitro and in vivo (see publication 2).
Studies behind Adaptos®
Additional publications
Veterinary orthopaedics
- DeVries WJ, Runyon CL, Martinez SA, Ireland WP. Effect of volume variations on osteogenic capabilities of autogenous cancellous bone graft in dogs. American Journal of Veterinary Research. 1996 Oct;57(10):1501-1505. PMID: 8896692.
- HOFFER, M.J., GRIFFON, D.J., SCHAEFFER, D.J., JOHNSON, A.L. and THOMAS, M.W. (2008), Clinical Applications of Demineralized Bone Matrix: A Retrospective and Case-Matched Study of Seventy-Five Dogs. Veterinary Surgery, 37: 639-647. https://doi.org/10.1111/j.1532-950X.2008.00430.x
Tissue engineering